Table of Contents
What Is Electric Charge?
It is essentially a feature inside the atom of matter. The charge is the electronic state of the object. When an atom accepts or excludes excess electrons, that state is called a charged atom.
The fine particles that make up atoms are called basic cells, they are electrons, protons, and neutrons.
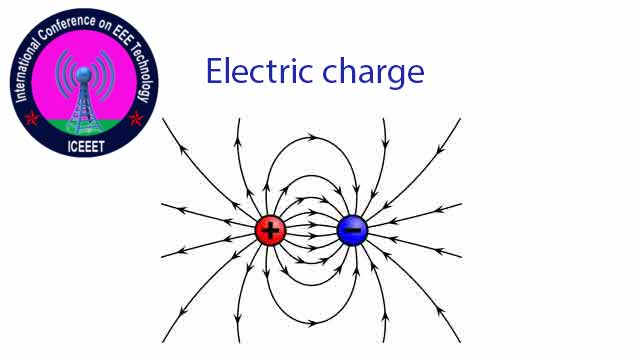
Electron exclusion or withdrawal is a positive charge (+ve) And a negative charge for receiving electrons (-ve) The unit of charge is Coulomb whom C Is published by
An electron is being charged = -1.602 x 10-19 C. Proton’s in charge = +1.602 x 10-19 C The value of the charge is always a whole number, eg 2e, 4e, 6e Etc.
The charge of the atom can be divided into two parts
- Carrier charges.
- Ionic charges.
The current that starts flowing due to rotation of a charge is called carrier charge, such as the hole, electron. As they move through the conductor, the current begins to flow.
Where there is an ionic charge, an electric field is formed around the atom. We know what the absence of electrons means in the hole and the charge and movement of electrons in conductive substances. And in the semiconductors, both the hole and the electrons move, so they charge and move.


1 comment
[…] Diode will keep the two current flow directions and will help with Charge and […]